 Wash the membrane (RNA side up) four times for 10 min each in 10 ml of wash buffer with shaking. Incubate the membrane (RNA side down) with an appropriate secondary IgG-HRP antibody diluted in blocking buffer for 1 h at room temperature with gentle shaking. Wash the membrane (flip the membrane so the RNA side faces up) three times for 10 min each in 10 ml of wash buffer with shaking. Incubate the membrane with antibody in 300 μl overnight at 4 degrees C with gentle shaking. Incubate each dilution with 1 μg/ml of the antibody you are testing. Wash the membrane in wash buffer for 10 min.ĭilute the nucleoside competitors serially in blocking buffer to give final concentrations of 1000/200/40/8/0 nM. Incubate the membrane (RNA side down) in 300 μl blocking buffer for 1 h at room temperature with gentle shaking. Wash the membrane in 500 μl of wash buffer for 5 min at room temperature with gentle shaking to wash off the unbound RNA. Remove the dish lid and crosslink the RNA to the membrane (RNA side up) with UV light: 125 mJoule/cm2 at 254 nM.Ĭut the membrane according to the grid and transfer to a clean 24 well plate. Transfer the dish with the membrane immediately into the chamber of SG Linker. Change tips after each loading, even between the same sample. Let the pipetted RNA droplet diffuse onto the membrane via surface tension. Note: Avoid touching the membrane with the pipette tip. An example assay map is given in appendix 1. Mix the RNA sample by pipetting up and down and drop 1 µL of RNA onto the p ositively charged nylon membrane.Transfer the membrane to a clean 10 cm plastic petri dish. Mark a grid on the membrane lightly with a pencil to guide sample loading. Cut the positively-charged membrane to an appropriate size. 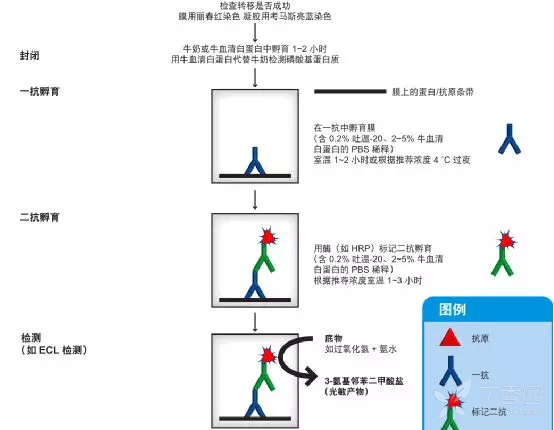
Chill the tubes on ice immediately after denaturation to prevent the re-formation of secondary structures of RNA.Denature the diluted RNA sample at 95 degrees in a heat block to disrupt secondary structures for 3 min.Dilute the RNA samples or oligos containing the modification of interest to an appropriate concentration with RNase free water.Methylene blue staining buffer (0.2% Methylene blue in 0.4M sodium acetate and 0.4M acetic acid).Secondary antibody (goat anti-rabbit IgG-HRP, ab97051, or goat anti-mouse IgG-HRP).Wash buffer, eg TBST (1x TBS, 0.1% Tween-20). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
